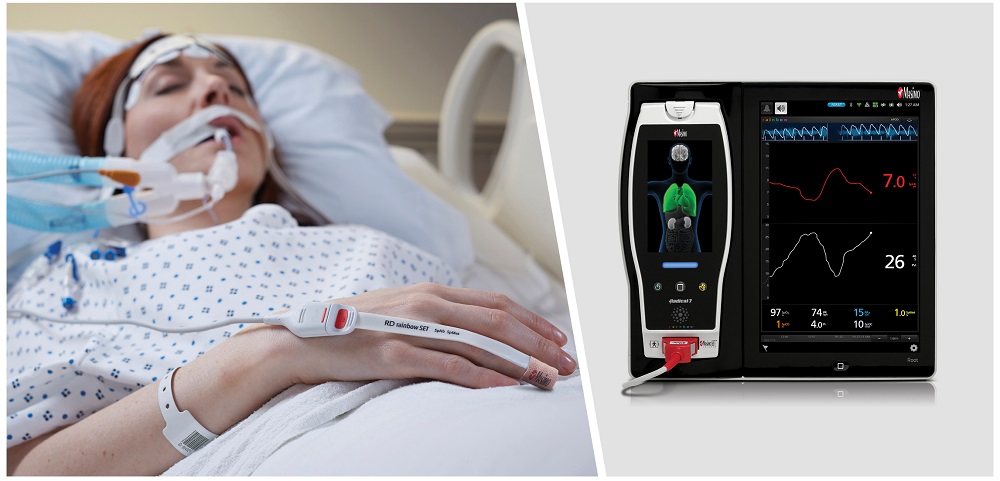
August 17, 2020 – Masimo (Irvine, CA) announced that PVi (pleth variability index) has received FDA clearance as a continuous, noninvasive, dynamic indicator of fluid responsiveness in select populations of mechanically ventilated adult patients. PVi, is a measure of the dynamic changes in perfusion index that occur during the respiratory cycle.
Available alongside Masimo SET pulse oximetry and rainbow Pulse CO-Oximetry on a variety of 2-LED, 4-LED, and 8-LED Masimo sensors, PVi is an index between 0 and 100 that is calculated using a proprietary algorithm based upon the relative variability of the pleth waveform.
Hospital protocols such as Enhanced Recovery After Surgery (ERAS) and Goal-directed Therapy (GDT) recommend fluid management as part of larger initiatives designed to improve patient care and safety. Fluid management protocols look to balance fluids by identifying when patients may be fluid responsive. Masimo says that the utility of PVi as a fluid responsiveness indicator has been demonstrated in more than 100 independent, published studies, including in ERAS and GDT protocols.
William Wilson, MD, MA, and chief medical officer at UCI Health, said, “Anesthesiologists and critical care physicians have long recognized the importance of dynamic measures of intravascular volume and fluid responsiveness. Previously this data could only be acquired using an invasive arterial line, and/or additional sophisticated devices. With the Masimo pleth variability index (PVi), one can now obtain this essential data using the pulse oximeter probe and following the continuous readout on the monitor. The PVi technology has undergone rigorous peer-reviewed evaluation, and demonstrated efficacy in determining adequacy of intravascular volume for guiding goal-directed therapy. The recent clearance of the Masimo PVi system will serve as another major breakthrough in promoting patient safety.”