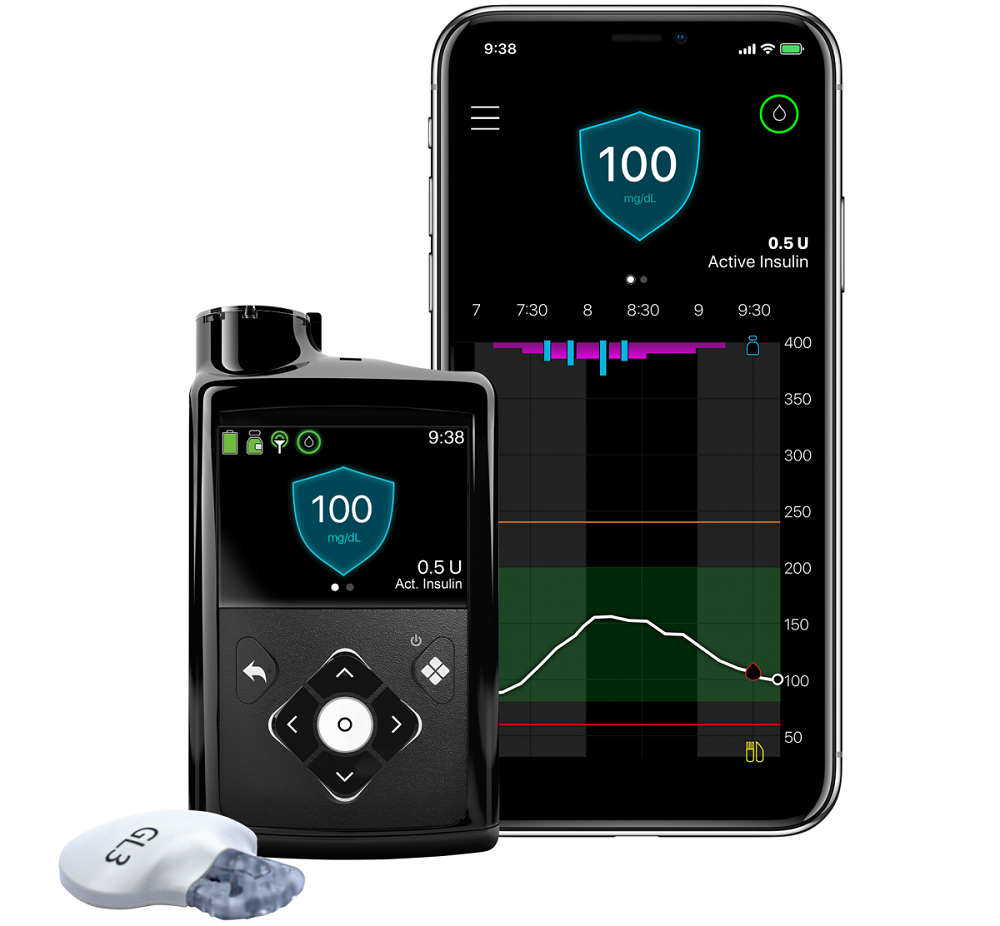
September 1, 2020 – Medtronic plc (Dublin, Ireland) received U.S. Food and Drug Administration (FDA) approval of its MiniMed 770G hybrid closed loop system. The new insulin pump system offers the company’s most advanced SmartGuard technology, as featured in the MiniMed 670G system, with the added benefits of smartphone connectivity and an expanded age indication to children as young as 2.
The company says that the new system expands the benefits of hybrid closed loop therapy to younger children living with Type 1 diabetes and makes it easier to access and share real-time CGM and pump data. The system will enable caregivers and care partners to see user data remotely on their smartphones, with proactive in-app notices sent when glucose levels are out of range. The data can also be shared automatically with clinicians and educators.
Medtronic says that the new connectivity gives the company the ability to provide upgrades to future technology via software updates which can further enhance security and device features.
Medtronic will begin taking orders for the new MiniMed 770G system in the U.S. this week.