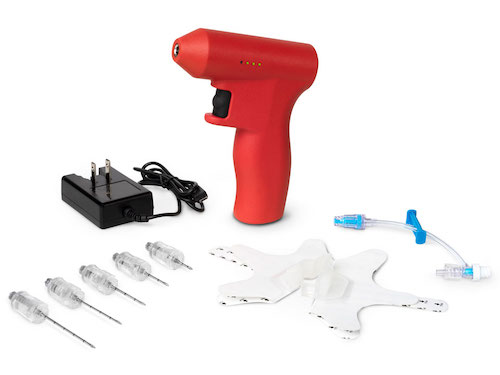
June 23, 2022 – BD announced a voluntary recall on the BD™ Intraosseous Needle Set Kits, BD™ Intraosseous Manual Driver Kits and BD™ Intraosseous Powered Drivers.
Certain lots within the expiration date of these intraosseous products may result in the following:
- Difficulty separating the stylet from the intraosseous needle, where increased force required to remove the stylet from the intraosseous needle results in inadvertent removal of the entire needle assembly during placement or inability to remove the stylet from an indwelling intraosseous needle, both resulting in functional loss of intraosseous access.
- Needle safety mechanism on the stylet not deploying post placement of the intraosseous needle and removal of the stylet from the needle.
- Metal discs intended to connect the needle assembly to the magnet in the powered driver sticking unexpectedly to the magnet, rendering the driver unable to be used.
These issues may result in delays in care due to limited or non-functioning intraosseous access or could also lead to needlestick injuries.
Customers should immediately review their inventory for the catalog and lot numbers listed above. Affected needle kits should be destroyed in compliance with the health care institution’s process for disposal. The use of affected intraosseous powered drivers should be paused until a BD representative provides instruction that it is safe for use. BD representatives will be reaching out to customers for inspection and will repair the devices if required.
There are no replacement products currently. BD recommends that customers evaluate their clinical needs and consider obtaining and using an alternative intraosseous product. BD will notify customers when replacement products become available.
